 To overcome this problem, Bohr assumed that electrons could only move in specific orbits, each one characterized by its energy level. Classical electromagnetism predicted that a charged particle moving in a circular way would emit energy so that the electrons should collapse on the nucleus in short moments of time. In this model the electrons revolve in circular orbits around the nucleus, occupying the lowest possible energy orbit, or the closest possible orbit to the nucleus. 
Bohr's atomic model was conceptually based on Rutherford's atomic model and the emerging ideas about quantization that had emerged a few years earlier with the research of Max Planck and Albert Einstein. He described the hydrogen atom as having a proton in the nucleus, and an electron revolving around it. With the discovery of this electron cloud, a more adequate model of the atom became available to scientists.īohr was trying to make an atomic model capable of explaining the stability of matter and the discrete emission and absorption spectra that are observed in gases. This particle became known as the neutron. James Chadwick interpreted this radiation as composed of particles with a neutral electrical charge and the approximate mass of a proton. Until 1932, it was believed that the atom was composed of a positively charged nucleus surrounded by negatively charged electrons. Where the cloud is denser, the probability of finding electrons is higher and, conversely, the electron is less likely to be in a less dense area of the cloud. This model can be represented as a nucleus surrounded by a subparticle. Unlike the Bohr model, this model does not define the exact path of an electron, but rather predicts the probabilities of the electron's location. This atomic model is known as the quantum mechanical model. In 1926, Erwin Schrödinger, an Austrian physicist, took Bohr's atomic model one step further. This model is based on the solar system and is known as the planetary model. He proposed that the electrons are arranged in concentric circular orbits around the nucleus. In 1913 Niels Bohr developed a new model of the atom. 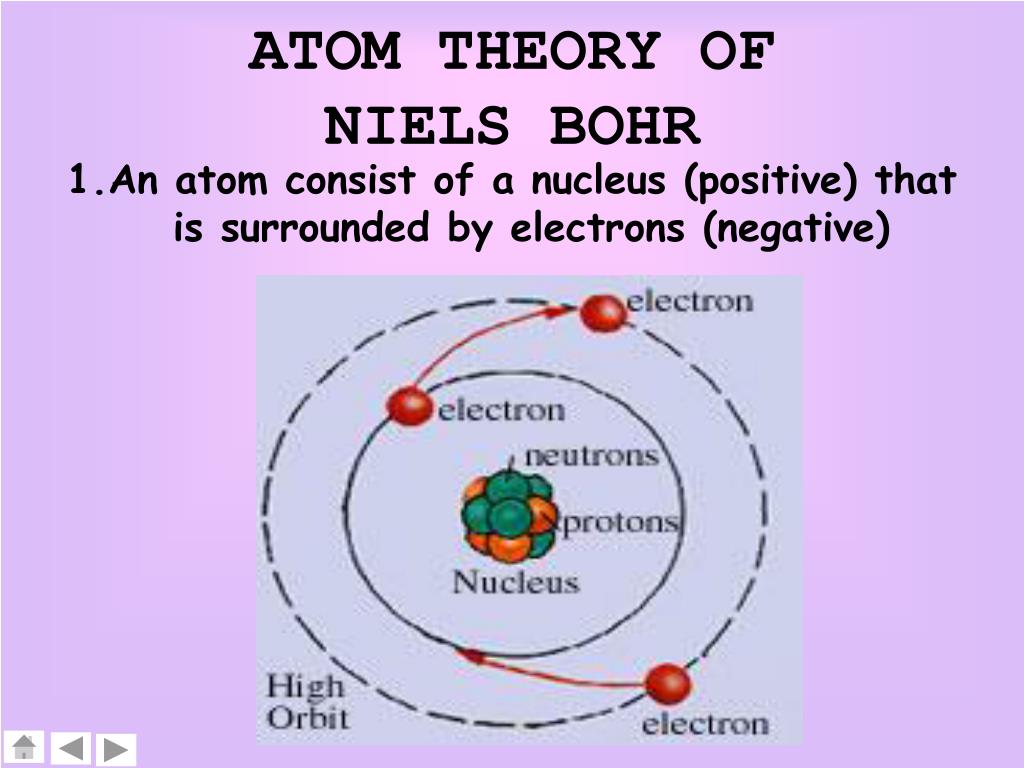
In addition, Bohr's model incorporated ideas taken from the photoelectric effect, explained by Albert Einstein. It was proposed in 1913 by the Danish physicist Niels Bohr, to explain how electrons can have stable orbits around the nucleus and why atoms exhibited characteristic emission spectra (two problems that were ignored in Rutherford's previous model). Since the quantization of momentum is adequately introduced, the model can be considered transactional in that it falls between classical and quantum mechanics. The Bohr model of the atom is a classical model of the atom, but it was the first atomic model in which it was proposed that electrons could only occupy specific orbits, called stable orbits. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
